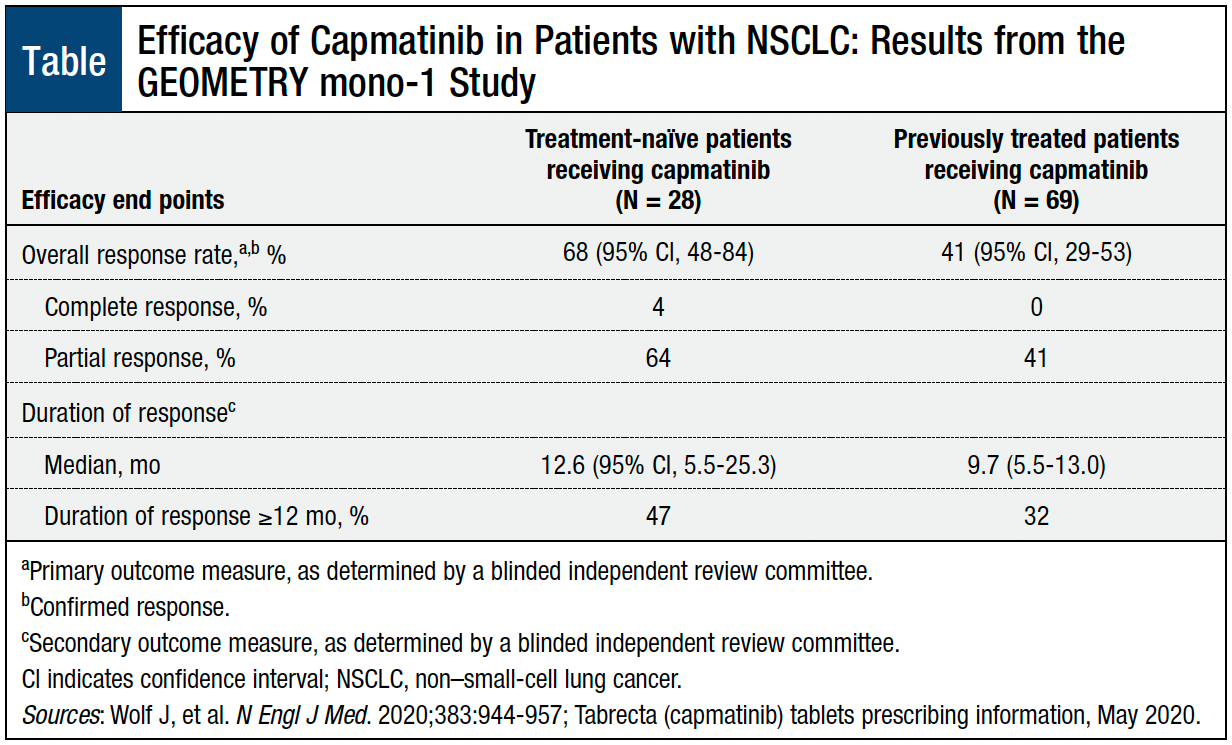
Tabrecta (Capmatinib) Approved for Patients with Metastatic NSCLC and MET Exon 14 Skipping Mutation | Oncology Practice Management
Real-world experience with capmatinib in MET exon 14-mutated non-small cell lung cancer (RECAP): a retrospective analysis from a

Phase 1 study of capmatinib in MET‐positive solid tumor patients: Dose escalation and expansion of selected cohorts - Bang - 2020 - Cancer Science - Wiley Online Library
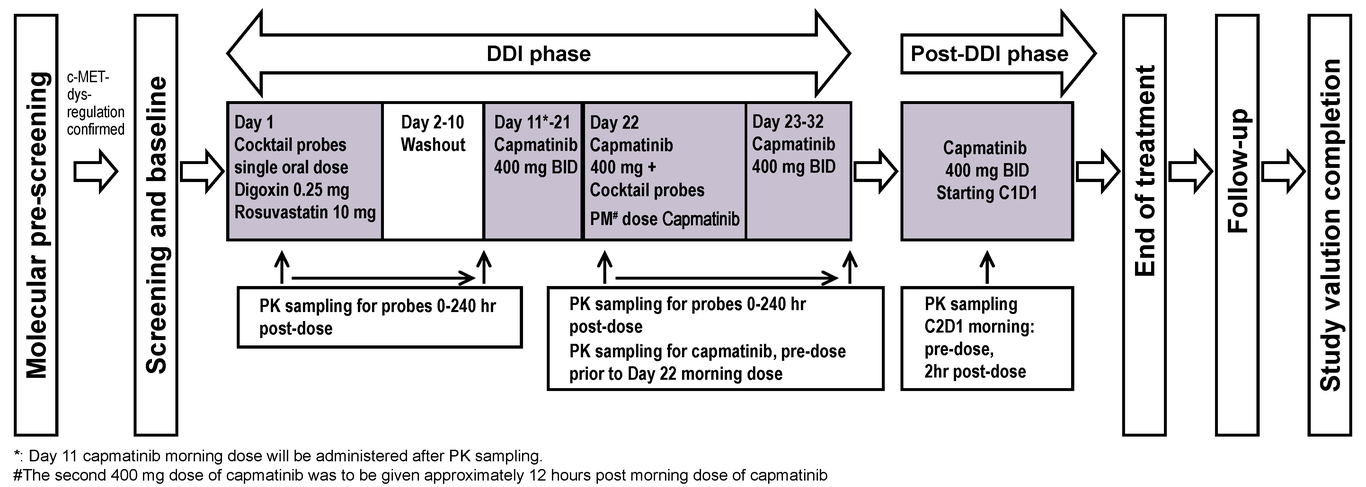
Effect of capmatinib on the pharmacokinetics of digoxin and rosuvastatin administered as a two-drug cocktail in patients with MET-dysregulated advanced solid tumors: A phase I, multicenter, open-label, single-sequence drug-drug interaction study -